New TEVAR devices
Published date : 27 April 2010
Article date : 27 April 2010
Several devices are currently available to treat thoracic pathologies, differing with respect to design, metallic composition and structure of the stent, and presence or absence of an active method of fixing the device to the aortic wall.
RESCUE trial: Valiant device
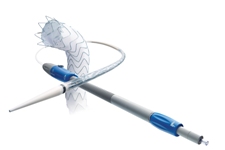 Launched in October, 2009, the Captivia Delivery System for the Valiant Thoracic Stent Graft (Medtronic) is a minimally invasive treatment for aneurysms and other lesions of the thoracic aorta. The Captivia Delivery System features tip capture for enhanced control of the stent graft during deployment and a hydrophilic coating applied to the graft cover to facilitate iliac access and delivery through patients’ vasculature.
Launched in October, 2009, the Captivia Delivery System for the Valiant Thoracic Stent Graft (Medtronic) is a minimally invasive treatment for aneurysms and other lesions of the thoracic aorta. The Captivia Delivery System features tip capture for enhanced control of the stent graft during deployment and a hydrophilic coating applied to the graft cover to facilitate iliac access and delivery through patients’ vasculature.
The Evaluation of the Clinical Performance of the Valiant Thoracic Stent Graft With the Captivia Delivery System for the Endovascular Treatment of Blunt Thoracic Aortic Injuries (RESCUE) study is currently recruiting patients and is expected to be completed by early 2011.The purpose of the study is to determine if the Valiant stent graft is safe and efficient in treating patients who have a blunt thoracic aortic injury (BTAI).
Primary outcome measures include all patients are alive (up to 30 days after implant) and secondary outcome measures are successful delivery and deployment of the stent graft, device, procedure and/or aortic related adverse events, and aortic related mortality (30 days after implant).
Patients included are those with a blunt thoracic aortic injury. Excluded patients include those who have received a previous stent or stent graft or previous surgical repair in the descending thoracic artery (DTA); have a history of bleeding diathesis, coagulopathy, or refuses blood transfusion; have a known allergy or intolerance to the device components; and have a known hypersensitivity or contraindication to anticoagulants or contrast media, which is not amenable to pre-treatment.
TX2
Cook’s new TX2 Pro-Form endograft, which has now obtained FDA approval, utilises an improved delivery system that allows for carefully controlled deployment of the endograft to help establish proximal conformity of the device to the aortic wall.
At the recent CX Vascular conference, London, UK, Mr Matt Thompson explained, “The challenges in the deployment are to achieve stability in high haemodynamic forces, attachment and sealing in proximal sealing zone, conformability to difficult angulated arch and long-term resistance to migration. If there was a fault with some of the early designs it was with conformability and achieving a seal in that angulated area just at the level of the left subclavian artery, the recent Proform modification that slightly deforms the proximal stent and allows it to make early contact with the aorta is a significant advantage.”
Furthermore, “the TX2 should be used particularly for the sort situation when you have a descending thoracic aneurysm finishing just between the level of the superior mesenteric artery and the coeliac access. We want to deploy at the level of the superior mesenteric artery and have distraction forces constrained by the axial fixation disc,” said Thompson.
TAG
Gore recently announced the first human implants of the next generation Conformable Gore TAG Thoracic Endoprosthesis in the US for the treatment of traumatic transection of the thoracic aorta. The commercially available GORE TAG Thoracic Endoprosthesis is a minimally invasive option for safely and effectively treating patients with descending thoracic aortic aneurysms. It is comprised of an ePTFE graft with an outer self-expanding nitinol support structure to combine both device flexibility and material durability. It received pre-market approval from the FDA in 2005.
In addition to traumatic aortic transection, the next generation Conformable GORE TAG Device has been approved to investigate endovascular repair of other etiologies including thoracic aortic aneurysm and aortic dissection.
The Conformable Gore TAG Device Traumatic Aortic Transection Trial will investigate the treatment of patients with aortic diameters of 16–42 mm.


