Categorised under:
Cardio-thoracic
>
Non-coronary intervention
>
Aortic Valves
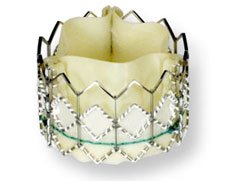
From Edwards Life Sciences
The Edwards SAPIEN transcatheter heart valve is an investigational device which is placed either through a transfemoral or transapical approach. The Edwards SAPIEN valve is being evaluated in the treatment of patients with severe calcific aortic stenosis who are considered to be high-risk or non-operable for conventional open-heart valve replacement surgery.
________________________________________________________________________________________________________________________________________________________________________

Our nominator said, "A disruptive technology that will change the treatment of aortic valvular disease"
Read the full list of Which Medical Device of the Year 2012 winners here.
________________________________________________________________________________________________________________________________________________________________________
 Which Medical Device of the Year 2011
Which Medical Device of the Year 2011
Runner-up: Cardiology (Jointly awarded with the CoreValve device)
"The judges couldn’t decide between these two devices, which have both stood 'the test of time'. The medium term (five years) performance of both valves is sufficiently robust to hope that TAVI may well become the first preferred option in patients with aortic stenosis." - Dr Azfar Zaman, Senior Editor for Cardiology. Read more
Your opinion matters to others - rate this device or add a comment

Did you know you can Register for FREE with this website?
Registration gives you full access to all of the features of WhichMedicalDevice. Find out more ...
WhichMedicalDevice is a FREE resource created by clinicians for clinicians.
Registration is free and gives you unlimited access to all of the content and features of this website.
Find out more...Registration is free and gives you unlimited access to all of the content and features of Which Medical Device. Find out more...
Which Medical Device is a community of clinicians sharing knowledge and experience of the devices and procedures we use on a daily basis. We ask that our members register with us so that we can maintain the unbiased and independent nature of our content. Registration is quick and free.
We do not make your details available to any third parties nor do we send unsolicited emails to our members. You can read our Privacy Policy here.